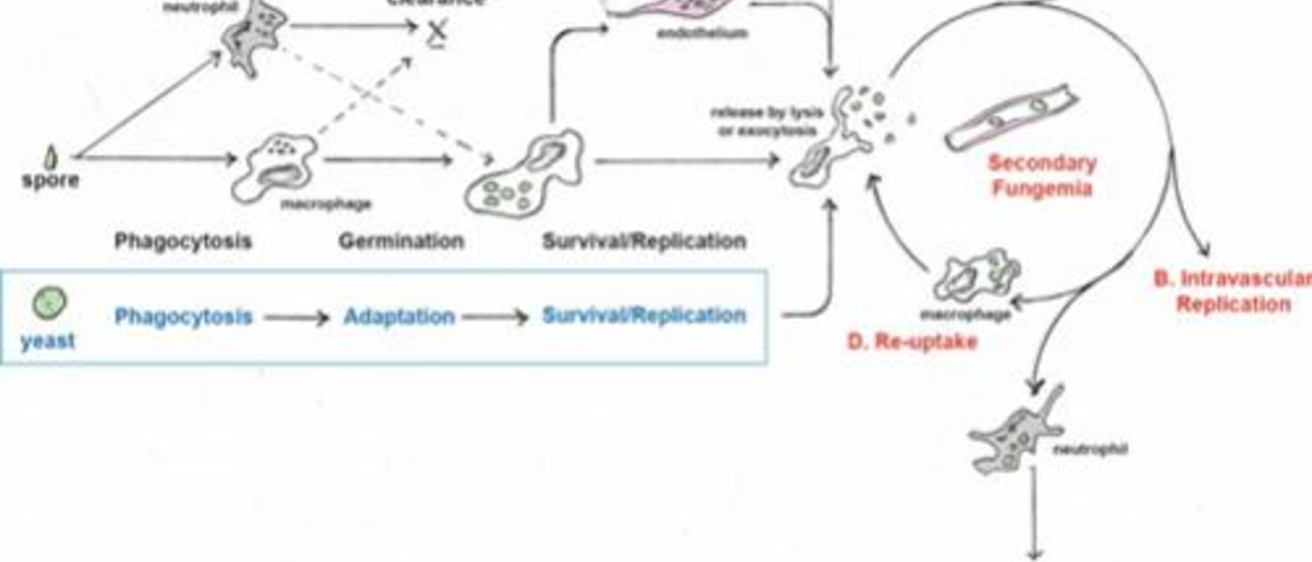
Cryptococcal meningoencephalitis is a fungal infection that predominantly affects immunocompromised patients and is uniformly fatal if left untreated. Timely diagnosis is difficult, and screening or prophylactic measures have generally not been successful. Thus, we need a better understanding of early, asymptomatic pathogenesis. Inhaled cryptococci must survive the host immune response, escape the lung, and persist within the bloodstream in order to reach and invade the brain. Here we took advantage of the zebrafish larval infection model to assess the process of cryptococcal infection and disease development sequentially in a single host. Using yeast or spores as infecting particles, we discovered that both cell types survived and replicated intracellularly and that both ultimately established a sustained, low-level fungemia. We propose that the establishment and maintenance of this sustained fungemia is an important stage of disease progression that has been difficult to study in other model systems. Our data suggest that sustained fungemia resulted from a pattern of repeated escape from, and reuptake by, macrophages, but endothelial cells were also seen to play a role as a niche for cryptococcal survival. Circulating yeast collected preferentially in the brain vasculature and eventually invaded the central nervous system (CNS). As suggested previously in a mouse model, we show here that neutrophils can play a valuable role in limiting the sustained fungemia, which can lead to meningoencephalitis. This early stage of pathogenesis-a balanced interaction between cryptococcal cells, macrophages, endothelial cells, and neutrophils-could represent a window for timely detection and intervention strategies for cryptococcal meningoencephalitis.
J. Muse Davis, Mingwei Huang, Michael R. Botts, Christina M. Hull and Anna Huttenlocher-----Infection and Immunity 2016